Carbohydrates consist of the elements carbon (C), hydrogen (H) and oxygen (O) with a ratio of hydrogen twice that of carbon and oxygen. Carbohydrates include sugars, starches, cellulose and many other compounds found in living organisms. In their basic form, carbohydrates are simple sugars or monosaccharides. These simple sugars can combine with each other to form more complex carbohydrates. The combination of two simple sugars is a disaccharide. Carbohydrates consisting of two to ten simple sugars are called oligosaccharides, and those with a larger number are called polysaccharides.
 Sugar and Cotton are Carbohydrates |
Sugars
Sugars are white crystalline carbohydrates that are soluble in water and generally have a sweet taste.
Monosaccharides are simple sugars
| Number of Carbons | Category Name | Examples |
|---|---|---|
| 4 | Tetrose | Erythrose, Threose |
| 5 | Pentose | Arabinose, Ribose, Ribulose, Xylose, Xylulose, Lyxose |
| 6 | Hexose | Allose, Altrose, Fructose, Galactose, Glucose, Gulose, Idose, Mannose, Sorbose, Talose, Tagatose |
| 7 | Heptose | Sedoheptulose |
Many saccharide structures differ only in the orientation of the hydroxyl groups (-OH). This slight structural difference makes a big difference in the biochemical properties, organoleptic properties (e.g., taste), and in the physical properties such as melting point and Specific Rotation (how polarized light is distorted). A chain-form monosaccharide that has a carbonyl group (C=O) on an end carbon forming an aldehyde group (-CHO) is classified as an aldose. When the carbonyl group is on an inner atom forming a ketone, it is classified as a ketose.
Tetroses
 |  |
| D-Erythrose | D-Threose |
Pentoses
 |  |  | 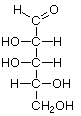 |
| D-Ribose | D-Arabinose | D-Xylose | D-Lyxose |
The ring form of ribose is a component of ribonucleic acid (RNA). Deoxyribose, which is missing an oxygen at position 2, is a component of deoxyribonucleic acid (DNA). In nucleic acids, the hydroxyl group attached to carbon number 1 is replaced with nucleotide bases.
 |  |
| Ribose | Deoxyribose |
Hexoses
Hexoses, such as the ones illustrated here, have the molecular formula C6H12O6. German chemist Emil Fischer (1852-1919) identified the stereoisomers for these aldohexoses in 1894. He received the 1902 Nobel Prize for chemistry for his work.
 |  | 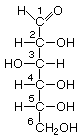 |  |
| D-Allose | D-Altrose | D-Glucose | D-Mannose |
 |  | 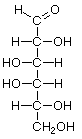 |  |
| D-Gulose | D-Idose | D-Galactose | D-Talose |
Structures that have opposite configurations of a hydroxyl group at only one position, such as glucose and mannose, are called epimers. Glucose, also called dextrose, is the most widely distributed sugar in the plant and animal kingdoms and it is the sugar present in blood as "blood sugar". The chain form of glucose is a polyhydric aldehyde, meaning that it has multiple hydroxyl groups and an aldehyde group. Fructose, also called levulose or "fruit sugar", is shown here in the chain and ring forms. The relationship between the chain and the ring forms of the sugars is discussed below. Fructose and glucose are the main carbohydrate constituents of honey.
 | 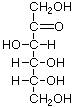 |  |  |  |
| D-Tagatose (a ketose) | D-Fructose | Fructose | Galactose | Mannose |
Heptoses
Sedoheptulose has the same structure as fructose, but it has one extra carbon. 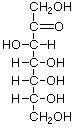 |
| D-Sedoheptulose |
Chain and Ring forms
Many simple sugars can exist in a chain form or a ring form, as illustrated by the hexoses above. The ring form is favored in aqueous solutions, and the mechanism of ring formation is similar for most sugars. The glucose ring form is created when the oxygen on carbon number 5 links with the carbon comprising the carbonyl group (carbon number 1) and transfers its hydrogen to the carbonyl oxygen to create a hydroxyl group. The rearrangement produces alpha glucose when the hydroxyl group is on the opposite side of the -CH2OH group, or beta glucose when the hydroxyl group is on the same side as the -CH2OH group. Isomers, such as these, which differ only in their configuration about their carbonyl carbon atom are called anomers. The little D in the name derives from the fact that natural glucose is dextrorotary, i.e., it rotates polarized light to the right, but it now denotes a specific configuration. Monosaccharides forming a five-sided ring, like ribose, are called furanoses. Those forming six-sided rings, like glucose, are called pyranoses.
 | 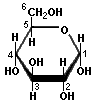 |  |  |
| D-Glucose (an aldose) | α-D-Glucose | β-D-Glucose | Cyclation of Glucose |
Stereochemistry
Saccharides with identical functional groups but with different spatial configurations have different chemical and biological properties. Stereochemisty is the study of the arrangement of atoms in three-dimensional space. Stereoisomers are compounds in which the atoms are linked in the same order but differ in their spatial arrangement. Compounds that are mirror images of each other but are not identical, comparable to left and right shoes, are called enantiomers. The following structures illustrate the difference between β-D-Glucose and β-L-Glucose. Identical molecules can be made to correspond to each other by flipping and rotating. However, enantiomers cannot be made to correspond to their mirror images by flipping and rotating. Glucose is sometimes illustrated as a "chair form" because it is a more accurate representation of the bond angles of the molecule. The "boat" form of glucose is unstable.
 | 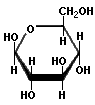 |  |
| β-D-Glucose | β-L-Glucose | β-D-Glucose (chair form) |
 |  |  |
| β-D-Glucose | β-L-Glucose | β-D-Glucose (boat form) |
Sugar Alcohols, Amino Sugars, and Uronic Acids
Sugars may be modified by natural or laboratory processes into compounds that retain the basic configuration of saccharides, but have different functional groups. Sugar alcohols, also known as polyols, polyhydric alcohols, or polyalcohols, are the hydrogenated forms of the aldoses or ketoses. For example, glucitol, also known as sorbitol, has the same linear structure as the chain form of glucose, but the aldehyde (-CHO) group is replaced with a -CH2OH group. Other common sugar alcohols include the monosaccharides erythritol and xylitol and the disaccharides lactitol and maltitol. Sugar alcohols have about half the calories of sugars and are frequently used in low-calorie or "sugar-free" products.
Xylitol, which has the hydroxyl groups oriented like xylose, is a very common ingredient in "sugar-free" candies and gums because it is approximately as sweet as sucrose, but contains 40% less food energy. Although this sugar alcohol appears to be safe for humans, xylitol in relatively small doses can cause seizures, liver failure, and death in dogs.
Amino sugars or aminosaccharides replace a hydroxyl group with an amino (-NH2) group. Glucosamine is an amino sugar used to treat cartilage damage and reduce the pain and progression of arthritis.
Uronic acids have a carboxyl group (-COOH) on the carbon that is not part of the ring. Their names retain the root of the monosaccharides, but the -ose sugar suffix is changed to -uronic acid. For example, galacturonic acid has the same configuration as galactose, and the structure of glucuronic acid corresponds to glucose.
 |  |  |
| Glucitol or Sorbitol (a sugar alcohol) | Glucosamine (an amino sugar) | Glucuronic acid (a uronic acid) |
Carbohydrates - Chemical Structure
Disaccharides consist of two simple sugars
| Disaccharide | Description | Component monosaccharides |
|---|---|---|
| sucrose | common table sugar | glucose 1α→2 fructose |
| maltose | product of starch hydrolysis | glucose 1α→4 glucose |
| trehalose | found in fungi | glucose 1α→1 glucose |
| lactose | main sugar in milk | galactose 1β→4 glucose |
| melibiose | found in legumes | galactose 1α→6 glucose |
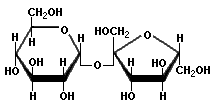 |  |  |
| Sucrose | Lactose | Maltose |
Sucrose, also called saccharose, is ordinary table sugar refined from sugar cane or sugar beets. It is the main ingredient in turbinado sugar, evaporated or dried cane juice, brown sugar, and confectioner's sugar. Lactose has a molecular structure consisting of galactose and glucose. It is of interest because it is associated with lactose intolerance which is the intestinal distress caused by a deficiency of lactase, an intestinal enzyme needed to absorb and digest lactose in milk. Undigested lactose ferments in the colon and causes abdominal pain, bloating, gas, and diarrhea. Yogurt does not cause these problems because lactose is consumed by the bacteria that transform milk into yogurt.
Maltose consists of two α-D-glucose molecules with the alpha bond at carbon 1 of one molecule attached to the oxygen at carbon 4 of the second molecule. This is called a 1α→4 glycosidic linkage. Trehalose has two α-D-glucose molecules connected through carbon number one in a 1α→1 linkage. Cellobiose is a disaccharide consisting of two β-D-glucose molecules that have a 1β→4 linkage as in cellulose. Cellobiose has no taste, whereas maltose and trehalose are about one-third as sweet as sucrose.
Trisaccharides
Raffinose, also called melitose, is a trisaccharide that is widely found in legumes and cruciferous vegetables, including beans, peas, cabbage, brussels sprouts, and broccoli. It consists of galactose connected to sucrose via a 1α→6 glycosidic linkage. Humans cannot digest saccharides with this linkage and the saccharides are fermented in the large intestine by gas-producing bacteria. Tablets containing the enzyme alpha-galactosidase, such as Beano, are frequently used as digestive aids to prevent gas and bloating. The enzyme is derived from selected strains of the food grade fungus Aspergillus niger.

Raffinose
Polysaccharides are polymers of simple sugars
Many polysaccharides, unlike sugars, are insoluble in water. Dietary fiber includes polysaccharides and oligosaccharides that are resistant to digestion and absorption in the human small intestine but which are completely or partially fermented by microorganisms in the large intestine. The polysaccharides described below play important roles in nutrition, biology, or food preparation.
Starch
Starch is the major form of stored carbohydrate in plants. Starch is composed of a mixture of two substances: amylose, an essentially linear polysaccharide, and amylopectin, a highly branched polysaccharide. Both forms of starch are polymers of α-D-Glucose. Natural starches contain 10-20% amylose and 80-90% amylopectin. Amylose forms a colloidal dispersion in hot water (which helps to thicken gravies) whereas amylopectin is completely insoluble.
- Amylose molecules consist typically of 200 to 20,000 glucose units which form a helix as a result of the bond angles between the glucose units.
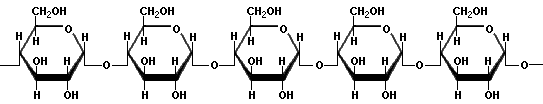
Amylose - Amylopectin differs from amylose in being highly branched. Short side chains of about 30 glucose units are attached with 1α→6 linkages approximately every twenty to thirty glucose units along the chain. Amylopectin molecules may contain up to two million glucose units.
 |
| Amylopectin |
 |
| The side branching chains are clustered together within the amylopectin molecule |
Starches are transformed into many commercial products by hydrolysis using acids or enzymes as catalysts. Hydrolysis is a chemical reaction in which water is used to break long polysaccharide chains into smaller chains or into simple carbohydrates. The resulting products are assigned a Dextrose Equivalent (DE) value which is related to the degree of hydrolysis. A DE value of 100 corresponds to completely hydrolyzed starch, which is pure glucose (dextrose). Dextrins are a group of low-molecular-weight carbohydrates produced by the hydrolysis of starch. Dextrins are mixtures of polymers of D-glucose units linked by 1α→4 or 1α→6 glycosidic bonds. Maltodextrin is partially hydrolyzed starch that is not sweet and has a DE value less than 20. Syrups, such as corn syrup made from corn starch, have DE values from 20 to 91. Commercial dextrose has DE values from 92 to 99. Corn syrup solids, which may be labeled as soluble corn fiber or resistant maltodextrin, are mildly sweet semi-crystalline or powdery amorphous products with DEs from 20 to 36 made by drying corn syrup in a vacuum or in spray driers. Resistant maltodextrin or soluble corn fiber are not broken down in the digestive system, but they are partially fermented by colonic bacteria thus providing only 2 Calories per gram instead of the 4 Calories per gram in corn syrup. High fructose corn syrup (HFCS), commonly used to sweeten soft drinks, is made by treating corn syrup with enzymes to convert a portion of the glucose into fructose. Commercial HFCS contains from 42% to 55% fructose, with the remaining percentage being mainly glucose. Modified starch is starch that has been changed by mechanical processes or chemical treatments to stabilize starch gels made with hot water. Without modification, gelled starch-water mixtures lose viscosity or become rubbery after a few hours. Hydrogenated glucose syrup (HGS) is produced by hydrolyzing starch, and then hydrogenating the resulting syrup to produce sugar alcohols like maltitol and sorbitol, along with hydrogenated oligo- and polysaccharides. Polydextrose (poly-D-glucose) is a synthetic, highly-branched polymer with many types of glycosidic linkages created by heating dextrose with an acid catalyst and purifying the resulting water-soluble polymer. Polydextrose is used as a bulking agent because it is tasteless and is similar to fiber in terms of its resistance to digestion. The name resistant starch is applied to dietary starch that is not degraded in the stomach and small intestine, but is fermented by microflora in the large intestine.
| fructose | 173 |
| invert sugar* | 120 |
| HFCS (42% fructose) | 120 |
| sucrose | 100 |
| xylitol | 100 |
| tagatose | 92 |
| glucose | 74 |
| high-DE corn syrup | 70 |
| sorbitol | 55 |
| mannitol | 50 |
| trehalose | 45 |
| regular corn syrup | 40 |
| galactose | 32 |
| maltose | 32 |
| lactose | 15 |
Glycogen
Glucose is stored as glycogen in animal tissues by the process of glycogenesis. When glucose cannot be stored as glycogen or used immediately for energy, it is converted to fat. Glycogen is a polymer of α-D-Glucose identical to amylopectin, but the branches in glycogen tend to be shorter (about 13 glucose units) and more frequent. The glucose chains are organized globularly like branches of a tree originating from a pair of molecules of glycogenin, a protein with a molecular weight of 38,000 that acts as a primer at the core of the structure. Glycogen is easily converted back to glucose to provide energy.
 |
| Glycogen |
Dextran
Dextran is a polysaccharide similar to amylopectin, but the main chains are formed by 1α→6 glycosidic linkages and the side branches are attached by 1α→3 or 1α→4 linkages. Dextran is an oral bacterial product that adheres to the teeth, creating a film called plaque. It is also used commercially in confections, in lacquers, as food additives, and as plasma volume expanders.
 |
| Dextran |
Inulin
Some plants store carbohydrates in the form of inulin as an alternative, or in addition, to starch. Inulins are present in many vegetables and fruits, including onions, leeks, garlic, bananas, asparagus, chicory, and Jerusalem artichokes. Inulins are polymers consisting of fructose units that typically have a terminal glucose. Oligofructose has the same structure as inulin, but the chains consist of 10 or fewer fructose units. Oligofructose has approximately 30 to 50 percent of the sweetness of table sugar. Inulin is less soluble than oligofructose and has a smooth creamy texture that provides a fat-like mouthfeel. Inulin and oligofructose are nondigestible by human intestinal enzymes, but they are totally fermented by colonic microflora. The short-chain fatty acids and lactate produced by fermentation contribute 1.5 kcal per gram of inulin or oligofructose. Inulin and oligofructose are used to replace fat or sugar and reduce the calories of foods like ice cream, dairy products, confections and baked goods.
 |
| Inulin n = approx. 35 |
Cellulose
Cellulose is a polymer of β-D-Glucose, which in contrast to starch, is oriented with -CH2OH groups alternating above and below the plane of the cellulose molecule thus producing long, unbranched chains. The absence of side chains allows cellulose molecules to lie close together and form rigid structures. Cellulose is the major structural material of plants. Wood is largely cellulose, and cotton is almost pure cellulose. Cellulose can be hydrolyzed to its constituent glucose units by microorganisms that inhabit the digestive tract of termites and ruminants. Cellulose may be modified in the laboratory by treating it with nitric acid (HNO3) to replace all the hydroxyl groups with nitrate groups (-ONO2) to produce cellulose nitrate (nitrocellulose or guncotton) which is an explosive component of smokeless powder. Partially nitrated cellulose, known as pyroxylin, is used in the manufacture of collodion, plastics, lacquers, and nail polish.
 |
| Cellulose |
Hemicellulose
The term "hemicellulose" is applied to the polysaccharide components of plant cell walls other than cellulose, or to polysaccharides in plant cell walls which are extractable by dilute alkaline solutions. Hemicelluloses comprise almost one-third of the carbohydrates in woody plant tissue. The chemical structure of hemicelluloses consists of long chains of a variety of pentoses, hexoses, and their corresponding uronic acids. Hemicelluloses may be found in fruit, plant stems, and grain hulls. Although hemicelluloses are not digestible, they can be fermented by yeasts and bacteria. The polysaccharides yielding pentoses on hydrolysis are called pentosans. Xylan is an example of a pentosan consisting of D-xylose units with 1β→4 linkages.
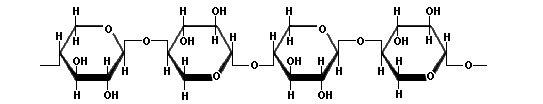 |
| Xylan |
Arabinoxylan
Arabinoxylans are polysaccharides found in the bran of grasses and grains such as wheat, rye, and barley. Arabinoxylans consist of a xylan backbone with L-arabinofuranose (L-arabinose in its 5-atom ring form) attached randomly by 1α→2 and/or 1α→3 linkages to the xylose units throughout the chain. Since xylose and arabinose are both pentoses, arabinoxylans are usually classified as pentosans. Arabinoxylans are important in the baking industry. The arabinose units bind water and produce viscous compounds that affect the consistency of dough, the retention of gas bubbles from fermentation in gluten-starch films, and the final texture of baked products.
 |
| Arabinoxylan |
Chitin
Chitin is an unbranched polymer of N-Acetyl-D-glucosamine. It is found in fungi and is the principal component of arthropod and lower animal exoskeletons, e.g., insect, crab, and shrimp shells. It may be regarded as a derivative of cellulose, in which the hydroxyl groups of the second carbon of each glucose unit have been replaced with acetamido (-NH(C=O)CH3) groups.
 |
| Chitin |
Beta-Glucan
Beta-glucans consist of linear unbranched polysaccharides of β-D-Glucose like cellulose, but with one 1β→3 linkage for every three or four 1β→4 linkages. Beta-glucans form long cylindrical molecules containing up to about 250,000 glucose units. Beta-glucans occur in the bran of grains such as barley and oats, and they are recognized as being beneficial for reducing heart disease by lowering cholesterol and reducing the glycemic response. They are used comercially to modify food texture and as fat substitutes.
 |
| Beta-Glucan |
Glycosaminoglycans
Glycosaminoglycans are found in the lubricating fluid of the joints and as components of cartilage, synovial fluid, vitreous humor, bone, and heart valves. Glycosaminoglycans are long unbranched polysaccharides containing repeating disaccharide units that contain either of two amino sugar compounds -- N-acetylgalactosamine or N-acetylglucosamine, and a uronic acid such as glucuronate (glucose where carbon six forms a carboxyl group). Glycosaminoglycans are negatively charged, highly viscous molecules sometimes called mucopolysaccharides. The physiologically most important glycosaminoglycans are hyaluronic acid, dermatan sulfate, chondroitin sulfate, heparin, heparan sulfate, and keratan sulfate. Chondroitin sulfate is composed of β-D-glucuronate linked to the third carbon of N-acetylgalactosamine-4-sulfate as illustrated here. Heparin is a complex mixture of linear polysaccharides that have anticoagulant properties and vary in the degree of sulfation of the saccharide units.
 |  |
| Chondroitin Sulfate | Heparin |
Agar and Carrageenan
Agar (agar agar) is extracted from seaweed and is used in many foods as a gelling agent. Agar is a polymer of agarobiose, a disaccharide composed of D-galactose and 3,6-anhydro-L-galactose. Highly refined agar is used as a medium for culturing bacteria, cellular tissues, and for DNA fingerprinting. Agar is used as an ingredient in desserts in Japan and other Asian countries. The gels produced with agar have a crispier texture than the desserts made with animal gelatin.
Carrageenan is a generic term for several polysaccharides also extracted from seaweed. Carrageenan compounds differ from agar in that they have sulfate groups (-OSO3-) in place of some hydroxyl groups. Carrageenan is also used for thickening, suspending, and gelling food products.
 Agarobiose is the repeating disaccharide unit in agar. |
Alginic acid, Alginates
Alginate is extracted from seaweeds, such as giant kelp (Macrocystis pyrifera). The chemical constituents of alginate are random sequences of chains of β-D-mannuronic and α-L-guluronic acids attached with 1→4 linkages. Alginates are insoluble in water, but absorb water readily. They are useful as gelling and thickening agents. Alginates are used in the manufacture of textiles, paper, and cosmetics. The sodium salt of alginic acid, sodium alginate, is used in the food industry to increase viscosity and as an emulsifier. Alginates are found in food products such as ice cream and in slimming aids where they serve as appetite suppresants. In dentistry, alginates are used to make dental impressions.

Alginic acid
Galactomannan
Galactomannans are polysaccharides consisting of a mannose backbone with galactose side groups. The mannopyranose units are linked with 1β→4 linkages to which galactopyranose units are attached with 1α→6 linkages. Galactomannans are present in several vegetable gums that are used to increase the viscosity of food products. These are the approximate ratios of mannose to galactose for the following gums:
- Fenugreek gum, mannose:galactose 1:1
- Guar gum, mannose:galactose 2:1
- Tara gum, mannose:galactose 3:1
- Locust bean gum or Carob gum, mannose:galactose 4:1
Guar is a legume that has been traditionally cultivated as livestock feed. Guar gum is also known by the name cyamopsis tetragonoloba which is the Latin taxonomy for the guar bean or cluster bean. Guar gum is the ground endosperm of the seeds. Approximately 85% of guar gum is guaran, a water soluble polysaccharide consisting of linear chains of mannose with 1β→4 linkages to which galactose units are attached with 1α→6 linkages. The ratio of mannose to galactose is 2:1. Guar gum has five to eight times the thickening power of starch and has many uses in the pharmaceutical industry, as a food stabilizer, and as a source of dietary fiber.
 Guaran is the principal polysaccharide in guar gum. |
Pectin
Pectin is a polysaccharide that acts as a cementing material in the cell walls of all plant tissues. The white portion of the rind of lemons and oranges contains approximately 30% pectin. Pectin is the methylated ester of polygalacturonic acid, which consists of chains of 300 to 1000 galacturonic acid units joined with 1α→4 linkages. The Degree of Esterification (DE) affects the gelling properties of pectin. The structure shown here has three methyl ester forms (-COOCH3) for every two carboxyl groups (-COOH), hence it is has a 60% degree of esterification, normally called a DE-60 pectin. Pectin is an important ingredient of fruit preserves, jellies, and jams.
 Pectin is a polymer of α-Galacturonic acid with a variable number of methyl ester groups. |
Xanthan Gum
Xanthan gum is a polysaccharide with a β-D-glucose backbone like cellulose, but every second glucose unit is attached to a trisaccharide consisting of mannose, glucuronic acid, and mannose. The mannose closest to the backbone has an acetic acid ester on carbon 6, and the mannose at the end of the trisaccharide is linked through carbons 6 and 4 to the second carbon of pyruvic acid. Xanthan Gum is produced by the bacterium Xanthomonas campestris, which is found on cruciferous vegetables such as cabbage and cauliflower. The negatively charged carboxyl groups on the side chains cause the molecules to form very viscous fluids when mixed with water. Xanthan gum is used as a thickener for sauces, to prevent ice crystal formation in ice cream, and as a low-calorie substitute for fat. Xanthan gum is frequently mixed with guar gum because the viscosity of the combination is greater than when either one is used alone.
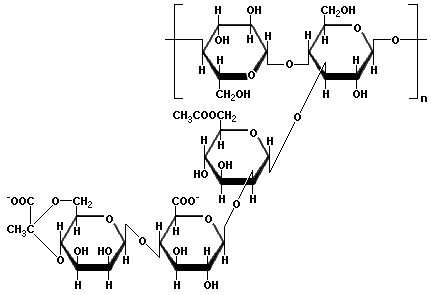 The repeating unit of Xanthan Gum |
Glucomannan
Glucomannan is a dietary fiber obtained from tubers of Amorphophallus konjac cultivated in Asia. Flour from the konjac tubers is used to make Japanese shirataki noodles, also called konnyaku noodles, which are very low in calories. Glucomannan is used as a hunger suppressant because it produces a feeling of fullness by creating very viscous solutions that retard absorption of the nutrients in food. One gram of this soluble polysaccharide can absorb up to 200 ml of water, so it is also used for absorbent articles such as disposable diapers and sanitary napkins. The polysaccharide consists of glucose (G) and mannose (M) in a proportion of 5:8 joined by 1β→4 linkages. The basic polymeric repeating unit has the pattern: GGMMGMMMMMGGM. Short side chains of 11-16 monosaccharides occur at intervals of 50-60 units of the main chain attached by 1β→3 linkages. Also, acetate groups on carbon 6 occur at every 9-19 units of the main chain. Hydrolysis of the acetate groups favors the formation of intermolecular hydrogen bonds that are responsible for the gelling action.

A portion (GGMM) of the glucomannan repeating unit.
The second glucose has an acetate group.
The main function is as a source of carbohydrates in food biokalori, besides that, also as a thickening or GMC in food technology as a stabilizer, sweetener (sucrose, glucose, fructose) and fuel, such as the glucose and starch, and as a constituent cell structure, such as cellulose and khitin. (Sudarmadji, 1996)
Carbohydrates have an important role in determining food characteristics such as taste, color and texture. While the function of carbohydrates in the body are:
1. Its main function as a source of energy (1 gram of carbohydrate produces 4 calories) for the needs of tissue cells of the body. Some of carbohydrates changed directly into energy for body activities, and partly in the form of glycogen stored in the liver and muscles. There are several body tissues such as the nervous system and red blood cells can only use the energy derived from carbohydrates alone.
2. Proteins to protect not burned as energy producers.
3. Body's need for energy is the first priority, when carbohydrate intake is insufficient for the body's energy needs and if there is not enough fat in the food or stored fat reserves in the body, it will replace the function of proteins as energy-producing carbohydrates. Thus the protein will leave its main function as a building material. If this situation continuous, then the state of energy and protein deficiency (KEP) can not be avoided anymore.
4. Helps metabolize fats and proteins, thereby preventing the occurrence of ketosis, and excessive protein breakdown.
5. In the liver function to detoxify toxic substances specific.
6. Several types of carbohydrates have a special function in the body. Lactose such as working to help the absorption of calcium. Ribose is an essential component in nucleic acids.
7. In addition some carbohydrate groups that can not be digested, contains fiber (dietary fiber) are useful for digestion, such as cellulose, pectin and lignin.
There are several classifications of carbohydrates, classification based on the body's ability to digest and classification based on a lot of sugar constituent. Based on the body's ability to digest carbohydrates, carbohydrates are classified into carbohydrates that can be digested and not digested. While classification based on the number of carbohydrates into sugar constituent monosaccharide, disaccharide, oligosaccharide and polysaccharide. While based on the complexity of late carbohydrates are divided into two simple kerbohidrat (Mono, Di, oligosaccharide) and complex carbohydrates (starch, Starch Non Polysacharide (NSP) and resistant starch).

source

SUMBER KARBOHIDRAT: Asupan makanan dengan kandungan karbohidrat tinggi seperti roti tawar, mie dan nasi sebaiknya dibatasi. Jika berlebih, bisa memicu serangan jantung.
 Subscribe
Subscribe
Copyright Violation! The text and images of this blog entry were copied without authorization from
BalasHapushttp://www.scientificpsychic.com/fitness/carbohydrates.html